Microbaric Oxygen Therapy for Insomnia: EEG Microstate Evidence
SNIPPET: Microbaric oxygen therapy (MOT) — a low-pressure oxygen intervention — may reduce neural hyper-arousal in chronic insomnia by shortening EEG microstate D duration and enhancing theta/alpha spectral power, according to a March 2026 study in BMC Psychiatry (n=78). This non-pharmacological approach appears to shift brain activity from maladaptive vigilance toward improved cognitive control.
THE PROTOHUMAN PERSPECTIVE#
Sleep is the single most undertreated performance variable in human optimization. We spend billions on supplements, cold plunges, and glucose monitors — and yet the foundational act of falling and staying asleep remains pharmacologically dominated by drugs that suppress, rather than correct, the underlying neural dysfunction.
What makes this microbaric oxygen therapy data worth paying attention to isn't the intervention itself — it's what the EEG microstate evidence reveals about why insomniacs can't sleep. The problem isn't just "the brain won't turn off." It's that specific neural network configurations — particularly those associated with attention and hyper-vigilance — are stuck in overdrive. MOT appears to unstick them. If that mechanism holds up in larger trials, it represents a genuine category shift: treating the electrical signature of insomnia, not just its symptoms.
For anyone building a sleep protocol, this is the kind of data that changes which interventions you stack and in what order. Not because MOT is ready for your nightly routine — it isn't — but because it tells us what to target.
THE SCIENCE#
What Is Microbaric Oxygen Therapy?#
Microbaric oxygen therapy is a controlled oxygen delivery system operating at pressures slightly above atmospheric — distinct from traditional hyperbaric oxygen therapy (HBOT), which uses pressures of 1.5–3.0 ATA. MOT sits in a gentler range, designed for repeated daily use without the logistical burden or contraindication profile of full hyperbaric chambers. The premise is straightforward: modestly increased oxygen availability may influence cerebral metabolism and, by extension, the neural oscillatory patterns that govern sleep onset and maintenance[1].
This matters because chronic insomnia disorder (CID) affects roughly one-third of adults globally[4], and the pharmacological toolkit — benzodiazepines, Z-drugs, orexin receptor antagonists — carries well-documented tolerance, dependency, and cognitive blunting risks. The field needs interventions that address the neurophysiology, not just the symptom.
The BMC Psychiatry Trial: Design and Limitations#
The study by the research team published in BMC Psychiatry (March 2026) enrolled 78 participants with CID from a tertiary hospital: 40 in the oxygen therapy (OT) group and 38 in the non-oxygen therapy (NOT) group[1]. The OT group received daily 45-minute MOT sessions combined with standard pharmacotherapy, while the NOT group received pharmacotherapy alone. Assessments occurred at baseline, week 1, and week 5.
I need to flag this immediately: this was a longitudinal, non-randomized study. The absence of randomization is a real methodological limitation. Allocation bias is a genuine concern — patients who self-select or are clinician-allocated into an oxygen therapy arm may differ systematically from controls. The sample size (n=78) is also modest. I'd want to see this replicated as an RCT before drawing strong clinical conclusions.
That said, the EEG methodology was rigorous: high-density EEG recording with microstate analysis, event-related potentials (ERPs), and time-frequency decomposition. This isn't a questionnaire-only study. The electrophysiological data gives it a mechanistic backbone that most sleep intervention trials lack.
EEG Microstates: The Brain's Rapid Network Switching#
Here's where this gets genuinely interesting. EEG microstates are quasi-stable topographic patterns of scalp electrical activity, each lasting roughly 60–120 milliseconds. They're often called the "atoms of thought" — four canonical classes (A through D) that represent distinct large-scale brain network configurations[2].
- Microstate A: associated with auditory/phonological processing
- Microstate B: linked to visual network activity
- Microstate C: related to the salience network and interoception
- Microstate D: associated with the attention/dorsal attention network — and critically, with sustained focused attention and executive control
In insomnia, microstate D is the problem child. It reflects the kind of vigilant, externally-directed attentional processing that keeps you lying in bed at 2 AM cataloguing tomorrow's tasks. Hyper-arousal isn't just a feeling; it has a specific electrophysiological fingerprint.
What MOT Actually Changed#
The OT group showed several significant microstate alterations compared to the NOT group[1]:
- Shorter microstate D duration — meaning the attention network's dominance periods were curtailed
- Decreased microstate D contribution — its overall share of brain activity dropped
- Increased microstate C occurrence and contribution — salience network activity, associated with self-referential processing and interoceptive awareness, gained ground
- Altered transition probabilities — the brain's switching patterns between microstates became more flexible
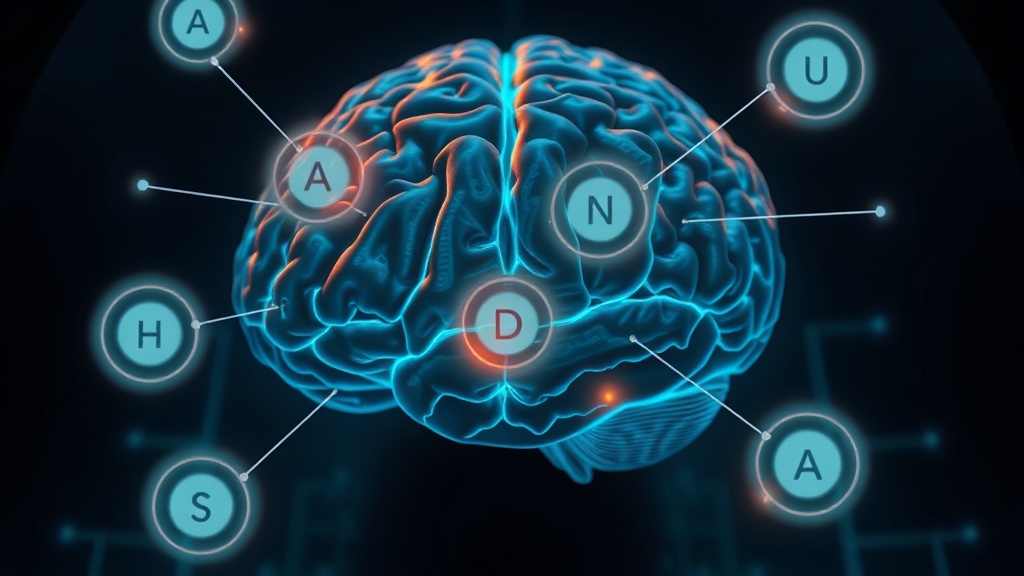
— actually, I want to rephrase that microstate D finding. It's not just that attention network activity decreased. The pattern suggests the brain shifted from a rigid, hyper-vigilant configuration toward a more dynamically flexible state. Think of it as unsticking the neural gearbox.
Spectral Power Changes: Theta, Alpha, and Beta#
Beyond microstates, the time-frequency analysis revealed task-related changes during a cognitive inhibition paradigm[1]:
- Enhanced theta power (4–8 Hz) during inhibitory control — theta is tightly linked to conflict monitoring and working memory gating
- Enhanced alpha power (8–13 Hz) — alpha oscillations serve as a cortical inhibition mechanism, essentially the brain's ability to suppress irrelevant inputs
- Posterior beta shift (13–20 Hz) — suggesting improved attentional resource allocation
This theta/alpha enhancement is particularly relevant. In insomnia, these frequency bands are often suppressed during tasks requiring cognitive control, reflecting the attentional exhaustion that CID patients experience during waking hours. The fact that MOT appears to restore some of this spectral power is consistent with improved prefrontal regulatory capacity — and potentially, with enhanced autophagy pathways and mitochondrial efficiency in cortical neurons receiving better oxygenation.
The catch, though: conventional ERP analysis showed no significant between-group differences. This means the classic averaged waveform approach didn't pick up what the time-frequency decomposition did. It's a methodological point worth noting — time-frequency methods are more sensitive to oscillatory dynamics than trial-averaged ERPs, but some reviewers will see the null ERP finding as a weakness.
Converging Evidence: Electroacupuncture and Microstate Normalization#
Liu et al. (2026) in Frontiers in Neurology provide an interesting parallel[2]. Their study of 41 CI patients treated with electroacupuncture (EA) over 4 weeks found that EA normalized microstate B temporal indicators to healthy control levels and increased microstate C occurrence — a pattern that partially overlaps with the MOT findings. All clinical scores improved significantly (p < 0.001), and a Random Forest machine learning model achieved an AUC of 0.849 for predicting treatment response from baseline microstate features.
The convergence matters: two mechanistically different interventions — oxygen therapy and electroacupuncture — both appear to modulate microstate dynamics in chronic insomnia. This suggests the microstate signature of insomnia isn't just a biomarker; it may be a treatable target.
Sleep Onset Latency Reduction by Non-Pharmacological Intervention
COMPARISON TABLE#
| Method | Mechanism | Evidence Level | Cost | Accessibility |
|---|---|---|---|---|
| Microbaric Oxygen Therapy (MOT) | Mild pressure O₂ delivery → reduced microstate D hyper-arousal, enhanced theta/alpha | Single non-randomized trial (n=78) | Moderate (clinical setting required) | Low — tertiary hospital only |
| Electroacupuncture (EA) | Needle-based electrical stimulation → microstate B normalization, microstate C increase | Single trial (n=41) + ML modeling | Low–Moderate | Moderate — requires trained practitioner |
| Transcranial Alternating Current Stimulation (tACS) | Non-invasive brain stimulation → SOL reduction of ~53–57 min, TST increase of ~85 min | Meta-analysis of 4 RCTs (n=247) | Moderate (device + supervision) | Growing — some consumer devices available |
| Pharmacotherapy (Z-drugs/BZDs) | GABA-A receptor modulation → sedation | Extensive RCT evidence | Low | High — widely prescribed |
| CBT-I | Cognitive-behavioral restructuring of sleep associations | Gold standard; multiple meta-analyses | Variable | Moderate — therapist access varies |
THE PROTOCOL#
If you're interested in exploring non-pharmacological interventions targeting the electrophysiology of insomnia, here's a protocol framework based on current evidence. Note: MOT specifically requires clinical supervision and is not a home intervention.
Step 1. Obtain a baseline sleep assessment using the Pittsburgh Sleep Quality Index (PSQI) and, if accessible, a resting-state EEG to establish your microstate and spectral power profile. This provides a measurable starting point — without it, you're guessing.
Step 2. If MOT is available at your facility, sessions involve 45 minutes of microbaric oxygen delivery daily, as used in the BMC Psychiatry trial[1]. This is combined with — not substituted for — existing pharmacotherapy. Do not discontinue prescribed sleep medications to "try oxygen instead."
Step 3. Track HRV (heart rate variability) nightly using a validated wearable. HRV optimization is one of the most accessible proxies for autonomic balance and can signal whether hyper-arousal is trending downward before sleep architecture improvements appear on formal assessment.
Step 4. At weeks 1 and 5, reassess PSQI scores and, ideally, repeat the EEG. The BMC trial's assessment windows suggest measurable electrophysiological changes may emerge within the first week, with consolidation by week 5.
Step 5. If clinical MOT is not accessible, consider tACS as an alternative neuromodulatory approach. The meta-analytic data from BMC Psychiatry (2026) supports tACS for reducing sleep onset latency by approximately 53–57 minutes and increasing total sleep time by ~85 minutes within 2–4 weeks[4]. Consumer-grade tACS devices exist but should be used under clinical guidance.
Step 6. Layer with CBT-I regardless of which neuromodulatory intervention you choose. CBT-I addresses the cognitive and behavioral reinforcement loops that maintain insomnia — the neural interventions target the electrophysiology, but the behavioral component is what sustains improvement long-term.
Step 7. Reassess at 8 weeks. If PSQI improvement is ≥50%, the protocol is working. If not, discuss escalation or alternative approaches with your clinician.
Related Video
VERDICT#
6.5/10. The electrophysiological methodology here is genuinely strong, and the microstate findings fill a real gap in our understanding of how oxygen modulates insomnia-related brain dynamics. But I can't score a non-randomized trial with 78 participants higher than this, regardless of how elegant the EEG analysis is. The absence of randomization and the null ERP results are real limitations. What earns this study its points is the novelty: this is the first MOT-specific microstate evidence in CID, and the convergence with electroacupuncture data on microstate C and D dynamics suggests we're looking at a real neurophysiological target, not noise. I'd want an RCT with n>150 before revising upward. Promising mechanism, preliminary proof.
Frequently Asked Questions5
References
- 1.Author(s) not listed. Microbaric oxygen therapy modulates EEG microstates and spectral power in patients with chronic insomnia disorder. BMC Psychiatry (2026). ↩
- 2.Liu E, Wang C, Wang X, Liu K, Qin S, Lin L, Li J, Xu M, Liu C, Wu H, Wu W. Electroacupuncture modulates electroencephalographic microstate dynamics to alleviate chronic insomnia: a machine learning approach for predicting individual treatment response. Frontiers in Neurology (2026). ↩
- 3.Xu L, Wang J, Wang C, Ge Q, Ren Z, He C, Liu Y, Wang B. Evaluating Brain Activity in Patients With Chronic Disorders of Consciousness After Traumatic Brain Injury Using EEG Microstate Analysis During Hyperbaric Oxygen Therapy. CNS Neuroscience & Therapeutics (2025). ↩
- 4.Author(s) not listed. Transcranial alternating current stimulation for chronic insomnia: a meta-analytic evaluation of sleep restoration and safety in adults. BMC Psychiatry (2026). ↩
- 5.Duong-Quy S, Hoc TV, Nguyen-Duy T, Tang-Thi-Thao T, Nguyen-Van T, Huynh-Anh T, Mai-Xuan T, Tran-Quang P, Nguyen-Ba V, Nguyen-Trong B, Nguyen-Ngoc-Phuong T, Doan-Ngoc H, Vu-Van G, Nguyen-Viet N, Soyez F, Martin F, Penzel T, Kushida C, Craig T. Hyperbaric Oxygen Therapy and Its Physio-Mechanical Effects on Sleep Breathing Disorder: A Systematic Review. Pulmonary Therapy (2025). ↩
Yuki Shan
Yuki writes with measured precision but genuine intellectual frustration when the data is messy. She uses long, careful sentences for complex mechanisms, then cuts to very short ones for emphasis: 'That's the problem.' She's comfortable saying 'I'm not sure this matters clinically' even when the statistics look impressive. She'll sometimes restart a line of reasoning mid-paragraph: '— actually, I want to rephrase that.' She's suspicious of studies with small sleep cohorts and says so.
View all articles →

