Heat Stress Nuclear Invagination: How Cells Reorganize Protein Metabolism
THE PROTOHUMAN PERSPECTIVE#
Your cells are not passive recipients of heat. They are architects.
When you step into a sauna at 80°C or sit in a hot bath pushing your core temperature up, your cells don't just produce heat shock proteins and call it a day. New research shows they physically reshape their nuclei — forming kidney-shaped invaginations that act as dedicated protein recycling stations. This is spatial intelligence at the subcellular level, and it changes how we should think about deliberate heat exposure.
For years, the biohacking community has anchored sauna protocols around HSP expression, cardiovascular adaptation, and growth hormone pulses. Those remain valid. But the discovery that heat triggers architectural reorganization of the nucleus — compartmentalizing where new proteins are built and damaged proteins are broken down — adds a layer of mechanistic depth that wasn't available six months ago. It means the cellular benefits of heat stress are more structurally coordinated than anyone assumed.
If you're using heat as a performance tool, you need to understand what's actually happening inside the cell. Not the marketing version. The real version.
THE SCIENCE#
Nuclear Invagination: Your Cells Reshape Under Heat#
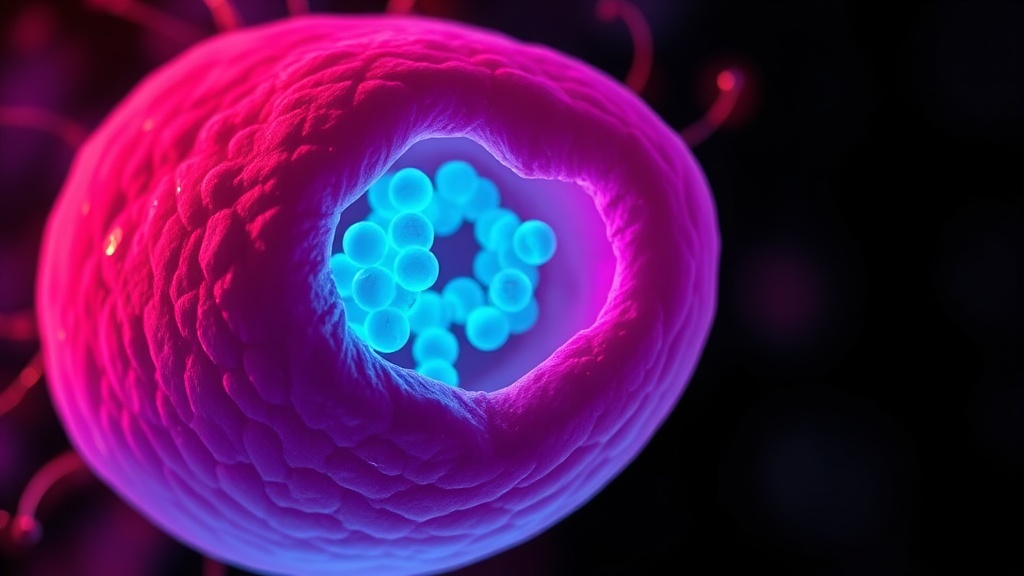
Nuclear invagination is the physical infolding of the nuclear envelope — the membrane surrounding the cell's nucleus — under stress conditions. Zhang et al. identified that heat stress produces reversible, kidney-shaped invaginations in the nuclear membrane, associated with the collapse of intermediate filaments and regional clustering of organelles around the invagination site[1].
This isn't cosmetic damage. These invagination regions function as specialized compartments where newly synthesized proteins are concentrated and protein degradation demand is heightened. Through immunofluorescence imaging and proteomic analysis, the research team demonstrated that cells effectively create localized "processing hubs" — spatial zones optimized for proteostasis under duress[1].
The critical detail: this compartmentalization correlates with differential heat tolerance across cell lines. Cells that form these invaginations more efficiently recover better from heat stress. Cells that don't form them as readily are more vulnerable. That's not a coincidence — it's a survival mechanism.
The Mechanosensor Gateway: How Cells Detect Heat#
But how does a cell even know it's hot? The answer, published in Nature Communications by a team studying budding yeast, points to a mechanosensor called Mid2 that detects heat-induced membrane stretch[3]. When Mid2 senses that the membrane is physically expanding from heat, it triggers rapid phosphorylation of the cytosolic Hsp70 chaperone Ssa1 at threonine 492 (T492).
This single phosphorylation event cascades into what the authors call an "epichaperome rearrangement" — a coordinated reshuffling of chaperone networks that fine-tunes translational pausing, HSF1 activity, MAPK signaling, and stress granule resolution[3]. It's not one pathway turning on. It's the entire proteostasis network recalibrating simultaneously, triggered by mechanical detection of heat at the membrane.
I find this particularly compelling because it reframes heat shock as a mechanically initiated event, not purely a thermochemical one. The membrane stretches. The sensor fires. Everything downstream follows.
Mitochondrial Thermogenesis: Heat From Within#
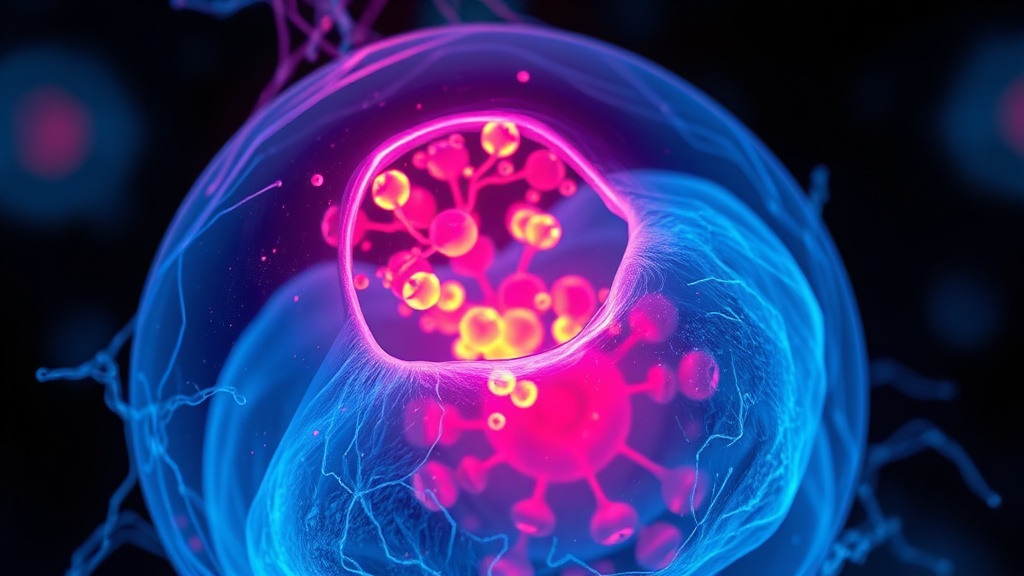
Here's where it gets complicated. Kang et al. demonstrated that cells don't need external heat to trigger the heat shock response — mitochondria can generate enough heat internally through the oxygen reduction reaction (ΔHf° = −286 kJ/mol) to activate HSF1 in the nucleus[6]. When mitochondrial uncoupling agents like FCCP drive proton import into the matrix, the resulting exothermic reaction produces heat that conducts to other organelles, including the nucleus.
This means mitochondrial thermogenesis itself can initiate nuclear stress granule formation and HSF1 chromatin localization — the same downstream events triggered by external heat[6]. The implication for anyone doing deliberate heat exposure is worth sitting with: your mitochondria are already producing heat signals that prime these pathways. External heat amplifies an existing intracellular communication channel.
I'm less convinced that the FCCP-mediated uncoupling model translates cleanly to what happens during sauna use in humans. The pharmacological uncoupling is more extreme than physiological thermogenesis. But the principle — that heat is a signaling molecule between mitochondria and the nucleus — is sound and has been validated with multiple fluorescent thermometers in mammalian cells[6].
Genome Restructuring and Nuclear Pore Dynamics#
The 3D architecture of the genome itself reorganizes under heat stress. Mohajan, Rubio, and Gross showed that nuclear basket proteins Nup2 and Mlp1, while dispensable for nuclear pore complex integrity, are required for driving heat shock response genes into coalesced chromatin clusters[2]. These proteins are recruited to HSR gene regulatory and coding regions in a heat shock-dependent manner.
In plain language: heat doesn't just turn genes on — it physically moves them closer together in 3D space to coordinate their expression. This genome restructuring happens downstream of transcriptional activation, meaning the cell first starts transcribing stress genes, then reorganizes the genome architecture to optimize continued expression[2].
Peroxisome Biogenesis: The Overlooked Organelle Response#
Shukla et al. added another piece to this puzzle by demonstrating that proteotoxic stress — including heat — drives peroxisome proliferation through activation of the heat shock response and inhibition of TOR signaling[5]. Peroxisomes, which mediate fatty acid oxidation and redox homeostasis, proliferate via increased de novo biogenesis from the endoplasmic reticulum.
Peroxisome biogenesis during heat stress is essential for cellular recovery, and this response was conserved across S. cerevisiae, K. phaffii, and primary human fibroblasts[5]. The TOR inhibition component is particularly relevant — it connects heat stress to the same nutrient-sensing pathway targeted by rapamycin and caloric restriction.
Exercise Plus Heat: The Combination Signal#
Normand-Gravier et al. tested what happens when you combine heat exposure (45 min at 40°C) with hybrid exercise in mice[4]. Key finding: while exercise alone increased proteosynthesis markers, the combination of exercise and heat produced the most pronounced upregulation of autophagy flux, evidenced by the LC3B-II/I ratio and P-NFκB levels[4].
The catch, though. At the 240-minute post-intervention timepoint, there were no additional effects of post-exercise heat on protein synthesis markers specifically. The combination drove autophagy and cellular stress markers higher — but not protein synthesis above exercise alone at that timepoint[4]. This suggests the heat-after-exercise window may be more relevant for cellular cleanup than for anabolic signaling.
Cellular Stress Responses: Exercise vs. Heat vs. Combined
COMPARISON TABLE#
| Method | Mechanism | Evidence Level | Cost | Accessibility |
|---|---|---|---|---|
| Sauna (80-100°C, 15-20 min) | HSF1 activation, HSP expression, nuclear invagination, autophagy upregulation | Multiple human observational + preclinical mechanistic studies | $0-30/session | High (gyms, spas, home units) |
| Hot water immersion (40-42°C) | Core temp elevation, HSP70 induction, cardiovascular adaptation | Replicated human trials | $0-5/session | Very high |
| Post-exercise heat (40°C, 45 min) | Enhanced autophagy flux, LC3B-II/I ratio increase, cellular stress amplification | Preclinical (mouse model) | $0-30/session | Moderate |
| Pharmacological HSP induction (e.g., celastrol) | Direct HSF1 activation without thermal stimulus | Preclinical only | Variable | Low (research compounds) |
| Mitochondrial uncoupling (endogenous) | Internal thermogenesis via ORR, HSF1 chromatin localization | Cell culture studies | N/A (endogenous) | N/A |
| Cold-to-heat contrast therapy | Alternating vasoconstriction/vasodilation, hormetic stress stacking | Limited human RCTs | $10-50/session | Moderate |
THE PROTOCOL#
Based on current evidence from the mechanistic studies above, here is a heat exposure protocol designed to maximize the newly identified nuclear compartmentalization and proteostasis responses. This is framed as exploratory — optimal parameters in humans are not yet established for these specific cellular endpoints.
Step 1. Pre-heat hydration and timing. Consume 500 mL of water 30-45 minutes before your heat session. If combining with exercise, complete your training first. The Normand-Gravier et al. data suggests post-exercise heat amplifies autophagy signaling more than either stimulus alone[4].
Step 2. Set your sauna to 80-85°C (dry) or use hot water immersion at 40-42°C. The mechanosensor-mediated response identified by the Nature Communications team requires sufficient membrane stretch — which means you need genuine core temperature elevation, not just skin warming[3]. Aim for a core temp increase of at least 1.0-1.5°C.
Step 3. Stay for 15-20 minutes in dry sauna, or 20-30 minutes in hot water immersion. The nuclear invagination response identified by Zhang et al. is reversible[1], which means it occurs during active heat stress and resolves during recovery. You need sustained exposure, not a quick dip. Start at 15 minutes, not 10. The adaptation response doesn't fully engage at lower durations based on HSP expression kinetics.
Step 4. Post-heat: do not immediately cold plunge. I know this is controversial in the biohacking community, and I say this as someone who loves cold exposure. But if your goal is to maximize the heat-induced proteostasis compartmentalization, immediate cold exposure may truncate the HSF1 activation window and stress granule resolution process[3]. Wait at least 20-30 minutes at room temperature before any cold intervention.
Step 5. Frequency: 3-4 sessions per week. The reversibility of nuclear invagination[1] suggests this is an acute adaptive response, not a permanent structural change. Regular repeated exposure likely trains the cellular machinery to execute this compartmentalization more efficiently — though I'd want to see longitudinal human imaging data before claiming that definitively.
Step 6. Track your recovery markers. HRV optimization the morning after heat sessions can serve as a proxy for systemic stress adaptation. If your HRV drops consistently post-sauna, you're likely overdoing frequency or duration. The cellular stress responses described in this research are protective when acute — but chronic overactivation of NFκB signaling (as seen in the combined exercise + heat condition) is not a goal[4].
Related Video
What is nuclear invagination and why does it matter for heat exposure?#
Nuclear invagination is the physical infolding of the nuclear envelope into a kidney-shaped structure during heat stress. Zhang et al. showed these invaginations create specialized compartments where protein synthesis and degradation are concentrated[1]. It matters because it reveals that heat exposure triggers architectural cellular changes — not just molecular ones — that directly support proteostasis and stress recovery.
How does the body detect heat at the cellular level?#
Cells detect heat through mechanosensors in the membrane. Research on the mechanosensor Mid2 showed it senses heat-induced membrane stretch and triggers phosphorylation of the Hsp70 chaperone Ssa1 at threonine 492[3]. This initiates a cascade that coordinates translational pausing, chaperone network reorganization, and stress granule dynamics. While this was demonstrated in yeast, the core sensing mechanisms are highly conserved.
Why might combining exercise with heat exposure enhance autophagy?#
Normand-Gravier et al. found that combining high-intensity exercise with 45 minutes of heat at 40°C produced the highest autophagy flux (LC3B-II/I ratio) compared to either stimulus alone in mice[4]. The likely explanation is that exercise creates misfolded proteins and cellular damage, while heat amplifies the cleanup machinery — particularly autophagy pathways and the LC3B processing system. However, this has not yet been replicated in human muscle tissue.
When should you avoid cold exposure after sauna?#
If your primary goal is maximizing HSF1-mediated heat shock protein expression and the nuclear compartmentalization response, immediate cold exposure may blunt these pathways by rapidly lowering core temperature before the stress response fully resolves. Based on the mechanistic data showing that stress granule resolution and epichaperome rearrangement require sustained signaling[3], waiting 20-30 minutes at ambient temperature before cold exposure is a reasonable precaution.
How does mitochondrial heat production relate to external heat stress?#
Kang et al. showed that mitochondria produce enough heat internally through the oxygen reduction reaction to activate HSF1 and initiate nuclear stress granule formation[6]. This means external heat exposure amplifies a signaling channel that already exists between your mitochondria and nucleus. The two sources of thermal stress — internal and external — converge on the same downstream pathways, potentially creating a synergistic effect.
VERDICT#
8/10. The Zhang et al. discovery of nuclear invagination as a protein metabolism compartmentalization strategy is genuinely novel — this is a mechanism that wasn't in anyone's model six months ago. The supporting evidence from the mechanosensor, mitochondrial thermogenesis, and TOR/HSR studies creates a coherent picture of how cells orchestrate their response to heat at multiple levels simultaneously. The exercise + heat combination data from Normand-Gravier et al. adds practical relevance, even if it's preclinical.
Where I dock points: nearly all of this is preclinical — yeast models, mouse models, cell cultures. The leap from kidney-shaped nuclear invaginations in cultured cells to "this is why your sauna protocol works" requires assumptions I'm not fully comfortable making yet. The human translation gap is real. But the mechanistic coherence across multiple independent labs and model organisms is strong enough that I'd bet heavily on these findings holding up as human studies catch up. If you're already doing deliberate heat exposure, this research validates the why at a deeper level than we've had before.
References
- 1.Zhang Z-H et al.. Heat Stress Triggers Nuclear Invagination and Spatial Compartmentalization of Protein Metabolism. Cell Proliferation (2026). ↩
- 2.Mohajan S, Rubio LS, Gross DS. Nuclear basket proteins Nup2 and Mlp1 drive heat shock-induced 3D genome restructuring downstream of transcriptional activation. Journal of Biological Chemistry (2025). ↩
- 3.Authors listed in Nature Communications. Mechanosensor-mediated Hsp70 phosphorylation orchestrates the landscape of the heat shock response. Nature Communications (2025). ↩
- 4.Normand-Gravier T, Solsona R, Arnould F, Deriaz R, Bertrand-Gaday C, Borrani F, Bernardi H, Sanchez AMJ. Acute effects of heat intervention and hybrid exercise on protein synthesis, ribosome biogenesis and autophagy. Journal of Thermal Biology (2025). ↩
- 5.Shukla N, Neal ML, Farré J-C et al.. TOR and heat shock response pathways regulate peroxisome biogenesis during proteotoxic stress. Nature Communications (2025). ↩
- 6.Kang M-G, Kim H-R, Lee HY, Kwak C, Koh H, Kang BH, Roe J-S, Rhee H-W. Mitochondrial Thermogenesis Can Trigger Heat Shock Response in the Nucleus. ACS Central Science (2024). ↩
Cira Renn
Cira writes with physical conviction — she's done this, she knows what it feels like, and she doesn't pretend otherwise. Her writing has visceral energy: 'Cold water at 10°C isn't a wellness trend. It's a physical confrontation.' She distinguishes between what the research shows and what she's experienced, and she'll tell you when they diverge.
View all articles →

