Vicarious Learned Helplessness: How Witnessing Stress Damages Your Brain
SNIPPET: Vicarious learned helplessness (VLH) is a new rodent model showing that merely witnessing another's trauma — without direct physical harm — produces depression-like pathology comparable to direct stress exposure. Kushwaha et al. (2026) found this "stress contagion" drives sex-dependent prefrontal cortex damage: males lose BDNF neurotrophic support while females show impaired oxytocin and synaptic scaffolding pathways.
THE PROTOHUMAN PERSPECTIVE#
We talk endlessly about optimizing our own stress response — cold plunges, adaptogens, HRV training. But what about the stress we absorb from other people? This research forces a question the biohacking community has largely avoided: can witnessing someone else's suffering physically alter your brain in ways that mirror direct trauma?
The answer, at least in mice, appears to be yes. And the damage isn't uniform — it splits along sex-dependent molecular lines in ways that should make anyone rethink the one-size-fits-all approach to stress resilience protocols.
For anyone working in high-empathy professions, doomscrolling through crisis media, or simply living with a partner who carries chronic stress, the implications are immediate. Stress contagion isn't metaphor. It's molecular. And the prefrontal cortex — the very region we rely on for executive function, emotional regulation, and decision-making — takes the hit differently depending on biological sex. If you're building a resilience stack without accounting for vicarious stress exposure, you may be missing the largest unaddressed variable in your protocol.
THE SCIENCE#
What Is Vicarious Learned Helplessness?#
Vicarious learned helplessness is a novel preclinical model in which an animal develops depression-like behavioral and neurobiological changes purely from observing another animal undergo inescapable stress. It matters because it isolates psychological stress contagion from physical harm — something traditional learned helplessness models have never cleanly separated. Kushwaha et al. (2026) found that observer mice developed behavioral symptoms "comparable to direct physical trauma" after just seven days of witnessing conspecifics receive foot shocks[1]. The model has already attracted attention among stress researchers studying empathy-driven psychopathology, and it builds on the broader framework described by Tafet and Ortiz Alonso (2025) connecting learned helplessness to prefrontal cortex dysfunction[3].
The Experimental Setup#
C57BL/6J mice — both male and female — were placed in partitioned chambers where they could see, hear, and smell a conspecific receiving inescapable foot shocks, but could not physically experience the shocks themselves. This multisensory but non-contact exposure continued for seven days. Post-conditioning, the observer mice underwent standard behavioral assays for anxiety and depression: active avoidance tests, sucrose preference (anhedonia), and elevated plus maze[1].
The result: vicarious stress was sufficient to produce the full depression phenotype. Active avoidance deficits, anhedonia, and anxiety-like behavior emerged in both sexes. Physiologically, both male and female observers showed HPA axis hyperactivity with elevated plasma corticosterone — the rodent equivalent of cortisol flooding.
I think the word "comparable" is doing a lot of heavy lifting here. The study claims VLH outcomes are comparable to direct stress, but without side-by-side effect sizes for every molecular marker, I'd want to see that quantified more precisely before fully endorsing the equivalence.
Sex-Dependent Prefrontal Cortex Pathology#
Here's where it gets complicated.
Both sexes shared some molecular damage in the prefrontal cortex: downregulation of metabotropic glutamate receptor 2 (mGluR2), which plays a critical role in modulating excitatory neurotransmission, and elevated interleukin-6 (Il6) mRNA, a proinflammatory cytokine linked to neuroinflammation and impaired autophagy pathways[1].
But the divergence was stark. Males showed specific deficits in BDNF (brain-derived neurotrophic factor) — both at the gene expression level (Bdnf) and protein level — along with reduced Nr3c1, the gene encoding the glucocorticoid receptor. This pattern suggests that vicarious stress in males attacks neurotrophic support systems and disrupts the cortisol feedback loop. Without adequate glucocorticoid receptor signaling, the HPA axis can't self-regulate — a molecular signature we see in treatment-resistant depression.
Females, by contrast, showed impaired oxytocin receptor expression (Oxtr) and reduced postsynaptic scaffolding proteins PSD-95 and SHANK3. The oxytocin finding is particularly interesting. Oxytocin isn't just a "bonding hormone" — it modulates GABAergic inhibition in the prefrontal cortex and shapes how social information gets processed. The loss of PSD-95 and SHANK3 suggests that synaptic architecture itself is degrading, which would impair synaptic plasticity and the kind of long-term potentiation needed for adaptive learning.
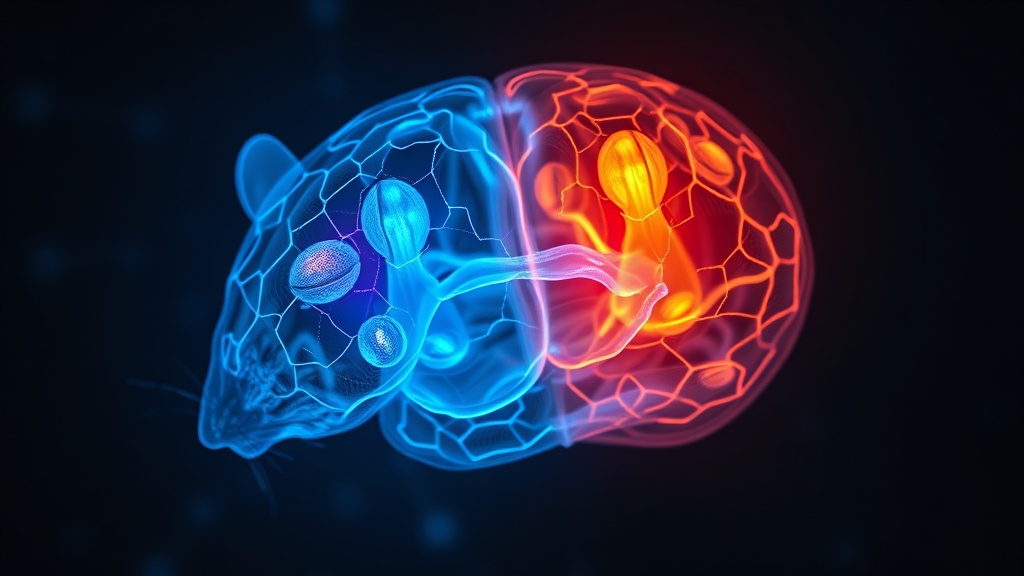
Convergence With Proteomics and Epigenetic Data#
This isn't happening in isolation. Irizarry-Méndez et al. (2026) identified 53 differentially expressed proteins in the infralimbic cortex of stress-susceptible female rats, including neurogranin and MAPT (microtubule-associated protein tau)[2]. Pathway enrichment analysis flagged alterations in synaptogenesis, clathrin-mediated endocytosis, calcium signaling, and chaperone-mediated autophagy — overlapping with several of the disrupted pathways in the VLH female mice. When they knocked down neurogranin or MAPT using AAV-shRNA in CaMKIIα-expressing neurons, extinction memory actually improved, suggesting these proteins may actively impair fear processing when overexpressed under stress.
Meanwhile, Domanico et al. (2025) showed that sub-chronic stress decreases global DNA methylation in the nucleus accumbens of both sexes, with sex-specific methylation changes at the prodynorphin and IKKβ gene sites[4]. The epigenetic dimension matters because it suggests these stress responses aren't just acute — they're written into the epigenome, potentially altering gene expression long after the stressor ends.
What does this actually feel like? I suspect the phenomenology of vicarious helplessness is different from direct trauma — more diffuse, harder to locate, maybe experienced as a vague emotional flatness rather than acute fear. But the molecular data suggests the prefrontal damage may be functionally equivalent. That gap between subjective experience and objective pathology is worth paying attention to.
Sex-Dependent Molecular Changes in Prefrontal Cortex After Vicarious Stress
COMPARISON TABLE#
| Method | Mechanism | Evidence Level | Cost | Accessibility |
|---|---|---|---|---|
| Vicarious Learned Helplessness (VLH) Model | Psychological stress contagion via multisensory witnessing; sex-dependent PFC pathology | Single preclinical study (mouse model) | Research-only | Lab setting only |
| Traditional Learned Helplessness | Direct inescapable physical stress (foot shock); serotonergic DRN/amygdala activation | Decades of replicated animal and human research | Research-only | Lab setting / clinical analogue |
| Single Prolonged Stress (SPS) | Acute multimodal physical stress; extinction memory deficits; proteomics-level changes | Multiple preclinical studies with proteomics validation | Research-only | Lab setting only |
| Sub-chronic Variable Stress | 5-day mild stressor paradigm; epigenetic changes (DNA methylation) in nucleus accumbens | Replicated in multiple labs | Research-only | Lab setting only |
| Learned Controllability Training | Perception of control → DL-PFC/VM-PFC activation; resilience building | Emerging clinical and preclinical evidence | Low (behavioral) | Clinical/self-directed |
THE PROTOCOL#
Based on the current evidence from Kushwaha et al. and the broader learned helplessness literature, here is a translational protocol for mitigating vicarious stress exposure. I want to be clear: this is extrapolated from preclinical data and existing clinical principles — not from a human trial of the VLH model itself.
Step 1: Audit Your Vicarious Stress Load. Track daily exposure to distressing content — news consumption, caregiving responsibilities, partner stress, workplace secondary trauma. Use a simple 1–10 distress scale logged twice daily (morning and evening) for two weeks. The data from Kushwaha et al. suggests that even passive, multisensory exposure to others' suffering drives measurable neurobiological change[1].
Step 2: Implement Deliberate Sensory Boundaries. The VLH model relied on multisensory cues — visual, auditory, olfactory. Reducing sensory channels of exposure may attenuate contagion. Practically: mute disturbing audio, limit doomscrolling to text-only consumption (no video), and create physical distance from chronically stressed individuals during your recovery windows.
Step 3: Target BDNF Support (Especially for Males). Given the male-specific BDNF deficits, prioritize interventions with established BDNF-boosting evidence: high-intensity interval exercise (shown to increase peripheral BDNF by 200–300% acutely), adequate sleep (7–9 hours — BDNF consolidation is sleep-dependent), and consider omega-3 fatty acid supplementation (2–3g EPA/DHA daily), which has shown BDNF-supportive effects in multiple human trials.
Step 4: Support Oxytocin and Synaptic Pathways (Especially for Females). The female-specific Oxtr and synaptic scaffolding deficits point toward interventions that enhance oxytocinergic signaling: positive physical touch, social bonding activities, and potentially magnesium supplementation (300–400mg glycinate or threonate daily), which supports NMDA receptor function and synaptic plasticity downstream of PSD-95.

Step 5: Train Learned Controllability. Tafet and Ortiz Alonso (2025) describe how perception of control activates the DL-PFC and VM-PFC while suppressing dorsal raphe nucleus and amygdala hyperactivation[3]. Practical application: daily micro-mastery tasks (small, completable goals), progressive skill challenges, and cognitive reappraisal exercises where you explicitly narrate control over one stressor per day. The point isn't to deny helplessness — it's to build the neural circuitry that counteracts it.
Step 6: Monitor HPA Axis Markers. Both sexes showed elevated corticosterone in the VLH model. In humans, track HRV as a proxy for autonomic balance (target rMSSD > 40ms for most adults), and consider morning salivary cortisol testing quarterly if you're in a high-vicarious-exposure role. Chronically elevated cortisol with suppressed HRV suggests your stress contagion load exceeds your recovery capacity.
Related Video
What is vicarious learned helplessness and how does it differ from standard learned helplessness?#
Vicarious learned helplessness is a model where an animal develops depression-like symptoms purely from witnessing another animal's inescapable stress, without experiencing physical harm itself. Traditional learned helplessness requires the subject to directly receive inescapable stress. The VLH model isolates psychological stress contagion as sufficient to produce comparable behavioral and molecular pathology[1].
Why do males and females show different brain changes from the same vicarious stress?#
The exact mechanisms aren't fully established, but Kushwaha et al. found that males lose BDNF and glucocorticoid receptor expression while females lose oxytocin receptor expression and synaptic scaffolding proteins (PSD-95, SHANK3) in the prefrontal cortex[1]. This likely reflects pre-existing differences in how male and female brains are wired for stress processing — different molecular substrates are more vulnerable depending on sex.
How can I tell if I'm experiencing stress contagion in daily life?#
There's no validated human diagnostic for vicarious learned helplessness yet. However, signs that map onto the preclinical findings include unexplained emotional flatness, reduced motivation (analogous to anhedonia), increased anxiety without personal threat, and difficulty with decision-making or executive function — all of which implicate prefrontal cortex function.
What is the role of mGluR2 in stress-related depression?#
Metabotropic glutamate receptor 2 modulates excitatory neurotransmission in the prefrontal cortex. Its downregulation, as seen in both male and female VLH mice, may contribute to glutamate excitotoxicity and impaired synaptic signaling. mGluR2 is an active drug target in depression research, with several agonists in clinical trials[1].
How does learned controllability help counteract helplessness?#
Learned controllability involves training the perception that one's actions can influence outcomes. Tafet and Ortiz Alonso (2025) showed this activates the dorsolateral and ventromedial PFC while suppressing amygdala and dorsal raphe nucleus hyperactivity — essentially reversing the neural signature of helplessness[3]. It can be cultivated through structured behavioral exercises and cognitive reappraisal.
VERDICT#
7.5/10. This is a genuinely novel preclinical model that addresses something the field has needed for years — a clean separation of psychological stress contagion from direct physical trauma. The sex-dependent molecular findings are the most valuable contribution, because they push back against unisex depression models that have dominated for decades. The limitation is obvious: it's a single study in mice. I'd want to see replication, dose-response curves for exposure duration, and — critically — any attempt to reverse the pathology pharmacologically before this shifts from "interesting model" to "actionable framework." The translational leap to humans is not yet justified, but the direction is right. If you're in a high-empathy profession or chronically exposed to others' distress, this data should at minimum make you take vicarious stress as seriously as your own direct stressors.
References
- 1.Kushwaha R, Kumar A, Chakravarty S, Soren K, Sinha D, Patel S. Vicarious learned helplessness: a translationally relevant novel model of stress contagion elucidating sex-dependent prefrontal cortex pathology. Frontiers in Behavioral Neuroscience (2026). ↩
- 2.Irizarry-Méndez N, Acosta-Pierantoni Y, Diaz-Vazquez A, Hernández A, Colón M, Tosado-Rodríguez EL, Cantres-Rosario YM, Roche-Lima A, Rodríguez-De Jesús AE, Meléndez LM, Porter JT. Proteomic insights into extinction memory deficits in stress-susceptible female rats. Frontiers in Behavioral Neuroscience (2026). ↩
- 3.Tafet GE, Ortiz Alonso T. Learned helplessness and learned controllability: from neurobiology to cognitive, emotional and behavioral neurosciences. Frontiers in Psychiatry (2025). ↩
- 4.Domanico MJ, Stevens S, Wainston I, Khoo E, McCall C, Swack BD, Sachs BD. Sub-chronic stress exerts partially distinct behavioral and epigenetic effects in male and female mice. Frontiers in Behavioral Neuroscience (2025). ↩
Fen Adler
Fen writes with psychological nuance and a slightly meandering quality that feels human. He'll start pursuing one idea, realize it connects to something else, and follow it briefly before returning: 'This reminds me of something from the attentional blink literature — different context, but the pattern holds.' He's interested in the experience, not just the mechanism, which means he'll occasionally ask: 'What does this actually feel like?' when discussing neurological effects.
View all articles →